Departments
Restorative
Sep 2007 —
Vol. 1,
Iss. 1
Adhesive Considerations in the Placement of Direct Composite Restorations
Abstract
The late Dr. John Gwinnett, one of the most brilliant
and respected members of the dental research and teaching community, often
characterized bonding as a chain, a series of links between the restorative
material being placed and the tooth tissues.1 And as in a chain,
the bond of a restorative material to the tooth
substrate is only as strong as its weakest link.
Dr. Gwinnett inspired a new generation of clinicians and researchers, the
author included, to think about adhesion in an entirely new fashion.
Insights and innovations by contemporaries, friends,
and colleagues of Dr. Gwinnett, including Byoung Suh, John Kanca, Charlie
Cox, Franklyn Tay, David Pashley, Sumita Mitra, Wayne Barkmeier, and many
others, also advanced our knowledge and understanding of adhesion. This
eventually led to the development of many of the various adhesive
techniques and materials utilized in dentistry today. This paper will focus on some of the current concepts and
techniques developed to manage the adhesive
interface during the placement of direct posterior composite restorations.
Direct composite resins
have the potential to offer a reasonably predictable alternative to amalgam
and other metal-based restoratives. This
assumes they are utilized in the appropriate clinical situation and are properly placed. In fact, the increasing demand for tooth-colored restorations, conservation of tooth structure, and
cosmetic dental procedures has encouraged the
widespread placement of direct composite
restorations.2-4 The greater level of clinical success
with direct composites is most likely related to
material developments, improved clinical skills and techniques, and dramatic advances in adhesive technology.5 Since the use of directly placed composites is a mainstay in the majority
of restorative practices, it is imperative
that dentists understand the rationale for
specific clinical techniques, as well as material idiosyncrasies, in order
to optimize the adhesive interface between the
composite restorative and the tooth substrate.
The majority of direct composites utilized in
restorative dentistry today consists of a methacrylated resin matrix (ie,
usually a blend of several resins) that is
mixed with various glass filler particles,
pigments, stabilizers, and chemical and/or light activated initiators. The filler particles in composites are typically
silanated. Silane serves as a coupling agent
between organics (ie, the resin matrix) and
inorganics (ie, the glass fillers). Filler particles can be manufactured in various shapes and sizes and from any number of inorganic glasses (eg, silica, zirconia-silica,
barium silicate, lithium, strontium, and
ytterbium). As a generalization, larger
particle, more heavily filled composites have superior physical properties while smaller particle composites, such as microfills, are not as strong but have a tendency to wear
and polish better.6 While an in-depth discussion and characterization of composite chemistry and classification is beyond the scope
of this particular
paper, clinicians should be aware that the specific nature of the composite being utilized
has a direct bearing on what is occurring at the adhesive interface. For example, all current
composite formulations shrink to some degree
during polymerization (ie, generally 1.5% to 5%
by volume). The total amount of shrinkage, the rate of shrinkage, and the
elastic modulus (ie, stiffness) of the composite are just some of the
factors that influence the degree of stress and
strain (ie, deformation) induced at the
adhesive interface during composite polymerization.
When placing a direct composite, the actual linkage
between composite and the tooth tissues is
usually mediated through the use of a dentin
bonding agent. The development of practical adhesive dentistry can be traced to Michael Buonocore who, in 1955, discovered he could increase the retention of acrylic-based
restoratives by first treating the teeth with
phosphoric acid.7 Subsequent research by Buonocore,
Gwinnett, and Matsui elucidated the mechanism of adhesion between enamel
and resin restoratives via resin tag formation.8 Long-term bonding
to phosphoric acid-etched enamel surfaces has
proven to be highly reliable and predictable; long-term bonding to dentin
is not as predictable, regardless of the dentin
bonding agent used. Clinicians can generally
bond predictably to enamel, but not nearly as predictably to dentin because of the morphologic, histologic, and compositional differences
between the two substrates.9 For one thing, dentin is a
highly variable substrate. Superficial, middle, and deep dentin can vary
significantly in their structural and chemical
composition. Enamel, on the other hand, is quite consistent throughout and
is also considerably more mineralized than dentin. The inorganic content of
mature enamel is approximately 96% hydroxyapatite by weight; the remainder
consists of water and organic material. Dentin,
on the other hand, is approximately 70%
hydroxyapatite by weight, 18% organic material (ie, predominantly
collagen), and 12% water.1,10 These percentages are not consistent and can vary
significantly depending on a number of factors,
including dentin depth, age of the teeth, and
history of tooth trauma and/or pathology. This, coupled with the relatively high water content of dentin, presents a
significant challenge for consistent and
reliable long-term bonding. However, this does not mean that stable and
strong initial
bonds to dentin cannot be attained. Laboratory
studies have shown that many current adhesive systems are capable of producing bond strengths to dentin that equal or surpass those of
acid-etched enamel controls.
The problem is that most of these are short-term
studies (ie, often 24 hours) and the focus needs to be on long-term
studies. It is of concern that the literature is replete with longer-term
studies—both in vitro and in vivo—that demonstrate a worrisome trend toward eventual degradation of the
dentin/adhesive interface.11-18 This could contribute
to the observed clinical problem of porcelain veneers sometimes debonding
over time when preparations are largely in dentin. Rarely is veneer
debonding a problem when significant amounts of enamel remain.19 Microleakage,
nanoleakage, hydrolysis, dentin permeability, pulpal pressure,
shrinkage stress, “water tree” formation, insufficient hybrid
layer formation, phase separation, dentin tubule orientation, occlusion,
enzymes released by bacteria, and operator error have all been implicated
as potential causes of deterioration of the dentin/adhesive interface over
time.20-25 When placing a direct composite, it makes sense to utilize
techniques and materials that, hopefully, will increase their long-term
predictability.
Know Your Adhesive System
All dentin bonding systems employ acids of one type or
another to facilitate adhesion to the tooth tissues. Acidic treatment of dentin and/or enamel creates a zone of demineralization,
which is subsequently (ie, total-etch) or
concurrently (ie, self-etch) infiltrated with various bifunctional primers
and resins. While many adhesive systems are capable of providing acceptable
clinical results if used in a knowledgeable fashion with attention to
detail, all have their particular idiosyncrasies.
The fourth generation, or three-step total-etch
systems,a-c generally have good long-term clinical track records and are
perhaps the most versatile of all the adhesive categories because they can be employed for
virtually any bonding protocol (ie, direct,
indirect, self-cure, dual-cure, light-cure).
These systems are still the “gold
standard” by which the newer systems are judged. Indeed, none of the newer systems in
the marketplace today perform any better, and often perform worse, than the
original multiple component total-etch systems of 15 years ago if bond strength to dentin/enamel, microleakage, and long-term
durability are used as the evaluation criteria.16
The fifth generation, or two-step total-etch systems,d-i evolved from the desire to simplify the three-step total-etch system
protocol. As a group, these are among the most
popular systems presently being utilized in
dentistry. They have generally proven to be highly effective, simpler, and faster than their multiple component predecessors. On the down side, many in this category, albeit
with some exceptions,
are not as predictable as the three-step total-etch systems when it comes to bonding to self- and dual-cure composites.26 In addition, the two-step total-etch systems may be more
susceptible to water degradation over time than three-step total-etch systems.27 This is because the polymerized primer of the two-step systems tends to be hydrophilic in nature. When using a three-step system, the
hydrophilic primer is covered by a more hydrophobic resin, making it
less susceptible to water sorption.21
If the clinician elects to utilize a fourth and fifth
generation total-etch system in the placement of a direct composite, he or
she needs to be aware that the majority of
laboratory studies show that these types of
systems perform best when placed on moist
dentin. This has been termed “wet”
bonding,28-30 although moist bonding may be a more accurate description of the phenomena. Dentin
exposed to phosphoric acid results in dissolution of the inorganic
hydroxyapatite matrix. As the matrix dissolves, the collagen fibrils, which are inherent in dentin, become exposed as they are no
longer supported and surrounded by their
inorganic scaffolding. It is this friable
“collagen network” that must be infiltrated by subsequently placed primers and resins to ensure good bonding. Air-drying
of acid-etched dentin causes collapse of the
collagen network and interferes with subsequent
primer/resin infiltration.31 In dentin that is left moist
(eg, after acid conditioning), the collagen fibrils remain in a relatively
“open” state and appear to be more permeable to subsequently placed primers and resins. The author’s recommended
technique when utilizing a total-etch protocol on unlined dentin is not to
air-dry the dentin once the phosphoric acid conditioner is washed off. The
excess water is simply blotted out with cotton pellets prior to placing the
primer. This results in a visibly moist dentin
surface instead of “puddles” of water, which should be avoided.32,33 It warrants noting that some total-etch systems, usually those that are acetone-based, appear to be more sensitive
to this wet/dry dentin issue than others (eg,
alcohol/water based systems). This highlights an inherent ambiguity many
have with the concept of wet bonding, namely
exactly how wet is wet?
Perhaps the biggest advantage of the sixth generation,
or two-component, self-etching systemsj-n is that their
efficacy appears to be less dependent on the hydration state of the dentin
than total-etch systems. Since the dentin is not pretreated with phosphoric
acid—as is the case with total-etch systems—no exposed collagen
layer is present to collapse on air-drying prior to placement of the
self-etching primers. Clinically this means that “wet” bonding
is not a concern and the tooth surface can be briefly air-dried prior to
placing a self-etching primer. This is not to say that self-etch systems
perform any better than total-etch systems, but they seem to be less
technique sensitive in this regard. One could also argue that a possible advantage of a self-etching system is that
demineralization of the dentin occurs concurrently with primer
infiltration. In
principle this helps ensure that the entire zone of demineralization is saturated with primer where it can then be polymerized in situ. On the down side, many products in this category do not etch enamel as well as their total-etch
cousins34 and many are not compatible with self- and dual-cure composites.26 A common clinical technique reported by many employing one of the
popular self-etching systemsj is to first etch the
enamel with traditional phosphoric acid prior to using it. This helps
ensure good bond strength to enamel but it does
require an additional step in the bonding
protocol. Those utilizing this technique should take care to confine the phosphoric acid solely to the enamel. Additional
etching of the dentin with phosphoric acid
could, in principle, create an “over-etch” situation where the
demineralization zone is too deep for subsequently placed primers to
completely penetrate.
The seventh generation, or one-bottle self-etching
systems,o-s represents the latest simplification of adhesive systems. With these systems all the ingredients required for bonding are
placed in, and delivered from, a single bottle.
This greatly simplifies the bonding protocol.
However, the price for simplification may be compromise. Incorporating and
placing all of the chemistry required for a viable adhesive system into a single bottle, and having it remain stable
over a reasonable period of time, poses a significant challenge. These inherently acidic systems tend to have a significant
amount of water in their formulations and may
be prone to hydrolysis and chemical breakdown.35,36 In addition,
once placed and polymerized, they are generally
more hydrophilic than two-step self-etching systems, which makes them more
prone to water sorption.37 This could contribute to hydrolysis and degradation of the
adhesive interface,38 as well as a reduction in mechanical properties of the
composite restorative.39 The acidic nature of the polymerized primers in seventh
generation adhesives generally makes them unsuitable
for use with self-cure composites since their acidic nature degrades the tertiary aromatic
amines required for chemical polymerization of
self-cure composites.37,40,41 It is this author’s opinion that while offering ease and simplicity,
seventh generation adhesive systems should be
used cautiously until more independent research clearly
demonstrates their short- and long-term effectiveness.
In principle, the “ideal” adhesive system
would be one that is hydrophilic when first placed in order to interact
with dentin, which inherently has a high water content, but then becomes
completely hydrophobic once polymerized in order to discourage water
sorption and hydrolysis. Unfortunately, no such chemistry currently exists.
A new, recently introduced total-etch systemt is among the first to address this issue by utilizing
chemistries that are less hydrophilic in nature.
Flowable Composite Liners
One common clinical technique thought to improve the
performance of direct composite restorations is
the use of flowable composites. With this
technique a flowable (ie, a lightly filled composite)
is placed in a thin layer after a dentin bonding agent is placed but prior to placement of
a more heavily filled composite restorative. In principle, flowable
composites, by virtue of their low viscosity, are able to get into the
“nooks and crannies” of the preparation. This helps ensure optimal adaptation to the previously placed
dentin bonding
agent, as well as to the higher viscosity composite restorative that is subsequently placed. One could also argue that
flowable composites, having a relatively low modulus of elasticity, are
able to act as stress-reducing liners during
polymerization and shrinkage of the
subsequently placed composite.42 The literature is
equivocal regarding the use of flowables under direct composite restorations in terms of
reducing microleakage, with some studies strongly supporting this technique
and others showing no benefit.43-50 Regardless of the research, many dentists have used
the technique with generally good success.
RMGI Liners
Resin modified glass ionomer (RMGI) liners represent an
alternative and, in this author’s
opinion, possibly better option to
flowable composite liners.51 With this technique a RMGI lineru is placed on the dentin
in a thin layer prior to placing a dentin bonding agent and composite restorative. RMGI
liners have several potential advantages over flowable composites. RMGI
liners have the intrinsic ability to both micromechanically and chemically
interact with dentin.52 They are simple to mix and place, release high sustained
levels of fluoride,53 have significant antimicrobial properties,54,55 evidence very
low solubility,56,57 and exhibit a favorable modulus
of elasticity and coefficient of thermal
expansion and contraction similar to that of dentin.58 One of the most important
characteristics of liners in regard to direct composites may be their
potential to act as stress-absorbing “buffers” during
polymerization shrinkage of the composite restorative. Both flowable
composite and RMGI liners have a low modulus of elasticity and the ability
to deform and/or flex to a degree when subjected to an external force. This
characteristic is thought to attenuate shrinkage stress from the subsequently placed higher modulus composite restorative
although, in the case of flowable composites,
at least one study showed this to be product specific.59 Another study showed that
when MOD composite restorations were placed in cavities lined with either a
flowable compositev or a RMGI linerw that the RMGI group had significantly less cusp deformation
due to polymerization shrinkage.60 This could be because the modulus of a RMGI is less than a
flowable, therefore more flexible, and/or the
fact that RMGI liners undergo hygroscopic expansion once polymerized, which may compensate for some of the polymerization shrinkage.61,62 In addition, the success of a flowable composite’s bond to dentin is highly contingent on the operator’s ability to first correctly place a dentin bonding
agent. A RMGI liner is placed prior to placing
a dentin bonding agent, making it much less technique sensitive in this
regard. Many clinicians have anecdotally reported a significant reduction
in postoperative sensitivity after switching to RMGI liners.62,63 The author
considers that the utilization of RMGI liners is also the simplest and most predictable method for managing microleakage/nanoleakage under direct composites. The literature is
replete with both in vivo and in vitro studies supporting this belief.64-76 Many of these studies directly
compare the use of flowable liners vs. RMGI liners in terms of controlling microleakage. The
author has been unable to find any study where
flowable composites, in conjunction with a
dentin bonding agent, outperform RMGI liners in this regard.
Don’t Forget the Small Details—WHICH
REALLY AREN’T THAT SMALL
• Check the expiration
date! Dentin bonding agents utilize chemistries that can deteriorate significantly over time.
Refrigeration may improve shelf-life,
but the dentin bonding agent should be removed
from the refrigerator 30 minutes prior to use because some may not perform
as well when cold.77
• Make sure to
evaporate the solvents. All adhesive systems
employ acetone, ethanol, water, or a
combination of these as solvents for their
particular monomers. It is very important to
evaporate these solvents by air-drying for an
adequate period of time prior to polymerization. Inadequate solvent evaporation results in incomplete resin polymerization and can
contribute to leakage and breakdown of the adhesive interface.78
• Periodically check
the bonding light. Light tips may become scratched or damaged, bulbs can lose intensity, and filters
can crack. All of these can significantly
reduce energy output. It is sound policy
to regularly test the energy output of bonding lights with a radiometer.
• If all else fails,
read the directions. Every adhesive system, even those in the same generation,
has specific placement idiosyncrasies that must
be precisely followed for optimal results. What
works well for one system may not be applicable for
another system.
The author’s personal preference in the
placement of direct composites is to utilize a
two-step total-etch systemd in conjunction with a RMGI
lineru.
The entire issue of wet bonding becomes
less of a concern with this technique because a significant portion of the exposed dentin is covered by the RMGI liner
prior to placing the phosphoric acid and resin primer.
The clinical technique is described below:
1. After the preparation is
complete, clean the dentin and enamel surfaces with slurry of fine pumice
and water using pumicex on a small brushy (Figures 1 View Figure and 2View Figure). The use of an antimicrobial solution is not recommended because in-house corporate studies have shown some of these solutions can have an
adverse effect on the adhesion of the
RMGI, which will be placed next.79 In any case, the
RMGI to be placed is antimicrobial.
2. Wash thoroughly with an air/water spray; quickly air-dry.
3. Mix and place a thin layer of the
RMGI lineru. Note: the author generally
covers most of the exposed dentin up to the dentoenamel junction (Figures 3 View Figure
and 4 View Figure). Light polymerize for 20 seconds. When
placing a RMGI line in this manner, the principle of “wet”
bonding using a total-etch system is no longer a major factor because most
of the dentin has already been covered by the RMGI.
4. Place phosphoric acid on all
enamel margins for 10 seconds; fill the entire
preparation with phosphoric acid for another 10 seconds (Figures 5 View Figure and 6 View Figure).
Wash thoroughly with air/water spray. Briefly air-dry. Blot drying is not required if most of the dentin has been covered by the RMGI.
5. Place several liberal coats of a two-step total-etch systemd (Figure 7 View Figure) and air-dry for a minimum of 10 seconds. Light-cure for 10 seconds.
6. Place a nanofilled compositez utilizing a
horizontal placement technique for the dentin
increments and a lateral placement technique
for the enamel increments (Figures 8 View Figure, 9 View Figure and 10 View Figure). Cure each increment for 10 seconds.
7. Finish and polish the completed
restoration (Figures 11 View Figure and 12 View Figure).
Conclusion
Proper management of the adhesive interface is crucial
for the predictable placement of direct
composites. This requires an understanding of
the materials being utilized, the substrate being bonded to, and a correct and precise clinical protocol. It is up
to clinicians to objectively examine their own clinical techniques, as well as successes and failures, in order to determine if
a change in materials and/or protocol is
warranted.
References
1. Gwinnett AJ. Bonding basics: What every clinician
should know. Esthetic Dent Update. 1994;5:35-41.
2. Cosmetic dental services expanding. Dent Prod Rep. 1998;1:34.
3. Allen EP, Brodine AH, Cronin RJ Jr, et al. Annual
review of selected dental literature: report of the Committee on Scientific
Investigation of the American Academy of Restorative Dentistry. J Prosthet
Dent. 2005;94:146-176.
4. Baratieri LN, Ritter AV. Four-year clinical
evaluation of posterior resin-based composite restorations placed using the
total-etch technique. J Esthet Restor Dent. 2001;13:50-57.
5. Alex G. Adhesive dentistry: where are we today? Compend
Contin Educ Dent. 2005;26:150-155.
6. Ferreira S, Kugel G, Martin S, et al. Direct
esthetic adhesive restorative materials. Inside Dentistry. 2006;2:
48-51.
7. Buonocore MG. A simple method of increasing the
adhesion of acrylic filling to enamel surfaces. J Dent Res. 1955;34:
849-853.
8. Buonocore MG, Gwinnett AJ, Matsui A. Penetration
of resin dental materials into enamel surfaces with reference to bonding. Arch
Oral Biol. 1968;13:61-70.
9. Alex G. Adhesive dentistry in the new millennium. Oral
Health. 2000;59-64.
10. Van Meerbeek B, Lambrechts P, Inokoshi S, et al.
Factors affecting adhesion to mineralized tissues. Oper Dent. 1992;Suppl 5:
111-124.
11. Hashimoto M, Ohno H, Kaga M, et al. Resin-tooth
adhesive interfaces after long-term function. Am J Dent. 2001;14:211-215.
12. Gwinnett AJ, Yu S. Effect of long-term water
storage on dentin bonding. Am J Dent. 1995;8:109-111.
13. Hashimoto M, Ohno H,
Kaga M, et al. In-vivo degradation of resin-dentin bonds in humans over 1 to 3 years. J Dent Res. 2000;79:1385-1391.
14. Meiers JC, Young D. Two-year composite/dentin
bond stability. Am J Dent. 2001;14:141-144.
15. Okuda M, Pereira PN, Nakajima M, et al.
Relationship between nanoleakage and long-term durability of dentin bonds. Oper
Dent. 2001:26;482-490.
16. De Munck J, Van
Landuyt K, Peumans M, et al. A critical review of the durability of adhesion to tooth tissue: methods and results. J
Dent Res. 2005;84:118-132.
17. Koshiro K, Inoue S, Tanaka T, et al. In-vivo
degradation of resin-dentin bonds produced by a self-etch vs. a total-etch
adhesive system. Eur J Oral Sci. 2004: 112:368-375.
18. Carrilho MR, Carvalho
RM, Tay FR, et al. Durability of resin-dentin bonds related to water and
oil storage. Am J Dent. 2005;18:315-319.
19. Christensen GJ. Are veneers conservative
treatment? J Am Dent Assoc. 2006; 137:1721-1723.
20. Tay FR, Frankenberger R, Krejci I, et al.
Single-bottle adhesives behave as permeable membranes after
polymerization. I. In vivo evidence. J Dent. 2004;32:611-621.
21. Hashimoto M, Ito S,
Tay FR, et al. Fluid movement across the resin-dentin interface during and
after bonding. J Dent Res. 2004;83:843-848.
22. Santerre JP, Shajii L, Leung BW. Relationship of
dental composite formulations to their degradation and release of
hydrolyzed polymeric-resin-derived products. Crit Rev Oral Biol Med. 2001;12:
136-151.
23. Pashley DH, Tay FR, Yiu C, et al. Collagen
degradation by host-derived enzymes during aging. J Dent Res. 2004;83:
216-221.
24. Finger WJ, Balkenhol
M. Practitioner variability effects on dentin bonding with an acetone-based one-bottle adhesive. J Adhes Dent. 1999;1:
311-314.
25. Purk JH, Dusevich V, Glaros A, et al. In vivo
versus in vitro microtensile bond strength of axial versus gingival cavity
preparation walls in Class II resin-based composite restorations. J Am Dent
Assoc. 2004;135:185-193.
26. CRA Newsletter. 2003;27(4):2-3.
27. De Munck J, Van Meerbeek B, Yoshida Y, et al.
Four-year water degradation of total-etch adhesives bonded to dentin. J
Dent Res. 2003;82:136-140.
28. Kanca J. Improving bond strength through acid
etching of dentin and bonding to wet dentin surfaces. J Am Dent Assoc. 1992;123:
35-42.
29. Kanca J. Effect of resin primer solvents and
surface wetness on resin composite bond strength to dentin. Am J Dent. 1992;5:
213-215.
30. Gwinnett AJ. Moist versus dry dentin: its effect
on shear bond strength. Am J Dent. 1992;5:127-131.
31. Gwinnett AJ. Chemically conditioned dentin: a
comparison of conventional and environmental scanning electron microscopy
findings. Dent Mater. 1994;10:150-155.
32. Tay FR, Gwinnett AJ, Wei SH. Micromorphological
spectrum from overdrying to overwetting acid-conditioned dentin in
water-free acetone-based, single-bottle primer/adhesives. Dent Mater. 1996;12:
236-244.
33. Pereira GD, Paulillo LA, De Goes MF, et al. How
wet should dentin be? Comparison of methods to remove excess water during
moist bonding. J Adhes Dent. 2001;3:257-264.
34. Pashley DH, Tay FR. Aggressiveness of
contemporary self-etching adhesives. Part II: etching effects on unground
enamel. Dent Mater. 2001;17:430-444.
35. Nishiyama N, Tay FR, Fujita K, et al. Hydrolysis
of functional monomers in single-bottle self-etching primer-correlation of
13C NMR and TEM findings. J Dent Res. 2006;85: 422-426.
36. Moszner N, Salz U, Zimmermann J. Chemical aspects
of self-etching enamel-dentin adhesives: a systematic review. Dent Mater. 2005;21:
895-910.
37. Tay FR, Pashley DH. Have dentin adhesives become
too hydrophilic? Can Dent Assoc. 2003;69:726-731.
38. Pashley DH. The evolution of dentin bonding. Dent
Today. 2003;22:112-114.
39. Bastioli C, Romano G, Migliaresi C. Water
sorption and mechanical properties of dental composites. Biomaterials. 1990;11;219-223.
40. Sanares AM, King NM, Itthagarun A, et al. Adverse
surface interactions between one-bottle light-cured adhesives and
chemical-cured composites. Dent Mater. 2001;17:542-556.
41. Suh BI, Feng L, Pashley DH, et al. Factors
contributing to the incompatibility between simplified-step adhesives and
chemically-cured or dual-cured composites. Part III. Effect of acidic resin
monomers. J Adhes Dent. 2003;5:267-282.
42. Terry DA, Leinfelder KF. Managing stress with
composite resin, Part 1. The restorative-tooth interface. Dent Today. 2006;25:
98-104.
43. Frankenberger R, Kramer N, Pelka M, et al.
Internal adaptation and overhang formation of direct Class II resin
composite restorations. Clin Oral Investig. 1999;3:208-215.
44. Chuang SF, Jin YT,
Liu JK, et al. Influence of flowable composite lining thickness on Class II composite restorations. Oper
Dent. 2004;29:301-308.
45. Perdigao J, Anauate-Netto C, Carmo AR, et al. The
effect of adhesive and flowable composite on postoperative sensitivity:
2-week results. Quintessence Int. 2004;35:777-784.
46. Attar N, Turgut MD, Gungor HC. The effect of
flowable resin composites as gingival increments on the microleakage of
posterior resin composites. Oper Dent. 2004;29:162-167.
47. Leevailoj C, Cochran MA, Matis BA, et al.
Microleakage of posterior packable resin composites with and without
flowable liners. Oper Dent. 2001;26:302-307.
48. Jain P, Belcher M. Microleakage of Class II
resin-based composite restorations with flowable composite in the proximal
box. Am J Dent. 2000;13:235-238.
49. Hagge M, Lindemuth JS, Mason JF, et al. Effect of
four intermediate layer treatments on microleakage of Class II composite
restorations. Gen Dent. 2001; 49:489-495.
50. Wibowo G, Stockton L. Microleakage of Class II
composite restorations. Am J Dent. 2001;14:177-185.
51. Alex G. The use of resin-modified glass ionomer
liners under composite resins: Should they be used to help control
microleakage? Inside Dentistry. 2005;1:30-33.
52. Yoshida Y, Van
Meerbeek B, Nakayama Y, et al. Evidence of chemical bonding at biomaterial-hard tissue
interfaces. J Dent Res. 2000;79:709-714.
53. Mitra SB. In vitro fluoride release from a
light-cured glass-ionomer liner/base. J Dent Res. 1991;70:75-78.
54. DeSchepper EJ, Thrasher MR, Thurmond BA.
Antibacterial effects of light-cured liners. Am J Dent. 1989;2:74-76.
55. Scherer W, Lippman N, Kaim J, et al.
Antimicrobial properties of VLC liners. J Esthet Dent. 1990;2:31-32.
56. Powell LV, Gordon GE, Johnson GH. Clinical
comparison of Class V composite and glass
ionomer restorations. Am J Dent. 1992;5:249-252.
57. Croll TP. Visible light-hardened glass-ionomer
cement base/liner as an interim restorative material. Quintessence Int. 1991;22:
137-141.
58. Mitra SB, Conway WT. Coefficient of thermal
expansion of some methacrylate modified glass
ionomers. J Dent Res. 1994;73:218. Abstract
#944.
59. Braga RR, Hilton TJ, Ferracane JL. Contraction
stress of flowable composite materials and their efficacy as
stress-relieving layers. J Am Dent Assoc. 2003; 134:721-728.
60. Alomari QD, Reinhardt JW, Boyer DB. Effect of
liners on cusp deflection and gap formation in composite restorations. Oper
Dent. 2001;26:406-411.
61. Leinfelder KF, Freedman G, Pakroo JS.
Postoperative sensitivity: bonded cavity liners revisited. Dent Today. 2001;20:
82-87.
62. Ruiz JL, Mitra S. Using cavity liners with direct
posterior composite restorations. Compend Contin Educ Dent. 2006;27:
347-351.
63. Christensen G. Reducing postoperative sensitivity
in Class I and Class II resin restorations. Dent Prod Report. 2001;February:
94-96.
64. Akpata ES, Sadiq W.
Post-operative sensitivity in glass-ionomer versus adhesive resin-lined
posterior composites. Am J Dent. 2001;14:34-38.
65. Sidhu SK, Henderson LJ. In vitro marginal leakage
of cervical composite restorations lined with a light-cured glass ionomer. Oper
Dent. 1992;17:7-12.
66. Gupta S, Khinda VI, Grewal N. A comparative study
of microleakage below cemento-enamel junction using light cure and
chemically cured glass ionomer cement liners. J Indian Soc Pedod Prev Dent. 2002;20:158-164.
67. Aboushala A, Kugel G, Hurley E. Class II
composite resin restorations using glass-ionomer liners: microleakage
studies. J Clin Pediatr Dent. 1996;21:67-70.
68. Dietrich T, Kraemer M, Losche GM, et al.
Influence of dentin conditioning and contamination of the marginal
integrity of sandwich Class II restorations. Oper Dent. 2000;25:
401-410.
69. Murray PE, About I, Franquin JC, et al.
Restorative pulpal and repair responses. J Am Dent Assoc. 2001;132:
482-491.
70. Beznos C.
Microleakage at the cervical margin of composite Class II cavities with different restorative techniques. Oper Dent. 2001;26:60-69.
71. Besnault C, Attal JP. Simulated oral environment
and microleakage of Class II resin-based composite and sandwich
restorations. Am J Dent. 2003;16:186-190.
72. Aboush YE, Torabzadeh H. Clinical performance of
Class II restorations in which resin composite is laminated over
resin-modified glass ionomers. Oper Dent. 2000;25:367-373.
73. Retief DH, McCaghren RA, Russell CM. Microleakage
of Vitrebond/P-50 Class II restorations. Am J Dent. 1992;5:
130-132.
74. Sidhu SK, Henderson L. Microleakage of cervical
composite restorations lined with light-cured glass ionomers. J Dent Res. 1990;69:
945. Abstract #102.
75. Loguercio AD, Alessandra R, Mazzocco KC, et al.
Microleakage in class II composite resin restorations: total bonding and
open sandwich technique. J Adhes Dent. 2002:4;137-144.
76. Guan J, Williams PT, Stockton L. Microleakage in
Class II composite restorations. J Dent Res. 2002:81 (Special Issue A):
Abstract #3360.
77. Sundfeld RH, da Silva AM, Croll TP, et al. The
effect of temperature on self-etching adhesive penetration. Compend Contin
Educ Dent. 2006;27:552-557.
78. Hashimoto M, Tay FR, Svizero NR, et al. The
effects of common errors on sealing ability of total-etch adhesives. Dent
Mater. 2006;22:560-568.
79. Mitra SB. Personal conversation.
Product Armamentarium
a All-Bond 2®, BISCO, Inc.,
Schaumburg, IL
b Scotchbond™ Multi-purpose
Plus, 3M™ ESPE™, St. Paul, MN
c OptiBond® FL, Kerr
Corporation, Orange, CA
d One-Step® Plus,
BISCO, Inc., Schaumburg, IL
e Adper™ Single Bond
Plus, 3M™ ESPE™, St. Paul, MN
f OptiBond® Solo, Kerr
Corporation, Orange, CA
g Prime&Bond® NT,
DENTSPLY Caulk, Milford, DE
h Excite®, Ivoclar
Vivadent, Amherst, NY
i One Coat® Bond,
Coltène/Whaledent, Cuyahoga Falls, OH
j Clearfil™ SE Bond,
Kuraray America, Inc., New York, NY
k Simplicity™, Apex Dental
Materials, Inc., Deer Park, IL
l All-Bond SE™ SPE,
BISCO, Inc., Schaumburg, IL
m Adper™ Prompt™ L-Pop™, 3M™ ESPE™, St. Paul, MN
n One Coat® Self-Etching
Bond, Coltène/Whaledent,
Cuyahoga Falls, OH
o I-Bond™, Heraeus
Kulzer, Armonk, NY
p G-Bond™, GC America,
Inc., Alsip, IL
q Xeno® IV,
DENSPLY Caulk Milford, DE
r Clearful® S3 Bond,
Kuraray America, Inc., New York, NY
s OptiBond® All-In-One,
Kerr Corporation, Orange, CA
t All-Bond 3™, BISCO, Inc.,
Schaumburg, IL
u Vitrebond™ Plus, 3M™ ESPE™, St. Paul, MN
v Revolution®, Kerr
Corporation, Orange, CA
w Vitrebond™, 3M™ ESPE™, St. Paul, MN
x Preppies™, Ultradent
Products, Inc., South Jordan, UT
y ICB® brush,
Ultradent Products, Inc., South Jordan, UT
z Filtek™ Supreme Plus,
3M™
ESPE™, St. Paul, MN
 |
 |
| Figure 1 Preoperative view. The
pre-existing restoration failed due
to recurrent decay. |
Figure 2 Fairly extensive decay
was removed and the preparation
cleaned with a wet pumice
mixture on a brush, after which
the preparation was washed and
briefly air-dried. |
| |
|
|
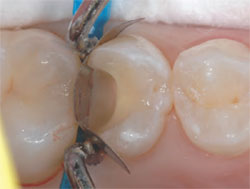 |
| Figure 3 A thin layer of the RMGI liner was dispensed, mixed, and placed with an applicator, after which it was light-cured for 20 seconds. A sectional matrix, wedge, and ring were also placed. |
| |
| Figure 4 Virtually all of the exposed dentin was covered with a thin layer of the RMGI liner, and a bevel of approximately 45° was placed on the enamel margins. |
| |
|
 |
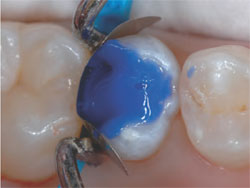 |
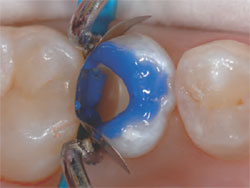
| Figures 5 and 6 Phosphoric acid was placed on all enamel margins for 10 seconds and then
flowed into the preparation for another 10 seconds. The acid was thoroughly washed out and
the preparation briefly dried. “Wet” bonding was not a significant issue because most of the
dentin was already covered with the RMGI liner. |
| |
|
 |
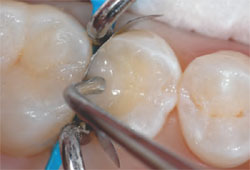 |
| Figure 7 Several coats of a dentin bonding agent were applied. It is very important to evaporate the solvent carriers prior to polymerization. |
| |
| Figure 8 The author utilized a horizontal
placement technique for the dentin increments.
A2 Body shade composite was
utilized for the dentin increments. |
| |
|
 |
 |
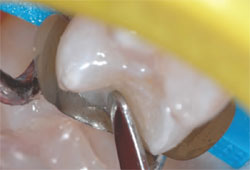
| Figure 9 A lateral placement technique
was utilized for enamel increments. The first
increment was pulled toward the buccal
margin, smoothed, and light polymerized.
A1 Enamel shade composite was used for
the enamel increments. |
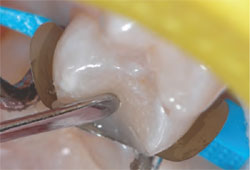
Figure 10 The next enamel increment
was added and pulled laterally toward the
lingual margin, smoothed, and light polymerized. |
| |
|
 |
 |
| Figure 11 Postoperative radiograph demonstrating proper gingival
embrasure form and what appears to be a well-integrated and
homogenous restoration. |
Figure 12 Immediate post-placement view of the case. Extrathin
finishing disks are essential for proper interproximal finishing
and polishing. |















